Why should you care whether you wash with real soap or a detergent cleanser? Aren’t they basically the same thing? Not quite. Products marketed as “soap” are not always true soap; many are actually synthetic detergent cleansers.1,2 That difference matters, because soaps and detergents are chemically different, regulated differently, and can raise different questions about skin tolerance, ingredient transparency, and overall health.
Published: Sept 2019 Last Updated: Apr 2026
Maybe you’ve wondered what the real differences are between soap and detergent. The terms are often used interchangeably, and most articles you’ll find online focus only on how well something cleans. But that’s only part of the story. To actually understand what you’re putting on your skin, you have to look at the chemistry behind these products, how they’re made, and—most importantly—what that means for safety and long-term exposure.
What’s the Difference Between Soap and Detergent?
At their core, both soaps and detergents are designed to do the same thing: remove dirt, oil, and debris from surfaces. They accomplish this because they are surfactants—molecules that allow oil and water to mix so contaminants can be rinsed away.3,4 But while they perform similar functions, the chemistry behind them is fundamentally different.
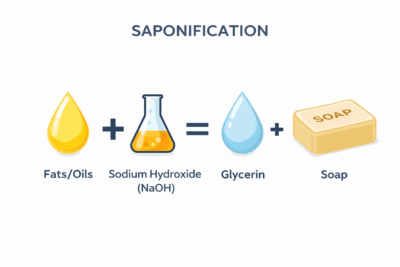
Soap is created through a process called saponification, where fats or oils react with a strong base such as sodium hydroxide or potassium hydroxide. The end result is a salt of a fatty acid that is what we commonly recognize as “real soap.”3 These fatty acid salts are effective cleansers, but they have a known limitation: they can react with minerals like calcium and magnesium in hard water, forming what is commonly referred to as “soap scum.”4
Detergents, on the other hand, are typically synthetic surfactants engineered to overcome some of soap’s limitations. Many are derived from petrochemical sources and are designed to remain effective in hard water without forming insoluble residues.3,4 This is one of the main reasons detergents became widely adopted, particularly in laundry applications where mineral buildup can affect both fabrics and machines.
It’s also important to correct a common misconception: most modern shampoos, body washes, and liquid “soaps” are not actually soaps at all—they are detergent-based formulations. These products rely on synthetic surfactants because they are easier to formulate, more stable, and perform consistently across a wide range of water conditions.
So while soaps and detergents may appear similar on the surface, they differ in how they are made, how they behave in real-world conditions, and what ingredients they introduce during use. And that distinction becomes especially important when we move beyond cleaning performance and begin to consider formulation complexity, ingredient transparency, and potential health implications.
How Is Real Soap Made?
Real soap is made through a chemical reaction known as saponification. In this process, fats or oils (which are chemically known as triglycerides) react with a strong base—typically sodium hydroxide (for solid bar soaps) or potassium hydroxide (for liquid soaps).5
During this reaction, the triglycerides are broken apart into two primary components: glycerin and fatty acid salts. These fatty acid salts are what we call “soap.” Each molecule has a dual nature—one end is attracted to water, while the other is attracted to oils and grease. This allows soap to lift and carry away dirt so it can be rinsed off with water.5,6
The specific properties of a soap depend heavily on the oils used to make it. Different fatty acids contribute different characteristics. For example, oils higher in saturated fatty acids tend to produce harder bars with more stable lather, while oils richer in unsaturated fatty acids generally create milder, more conditioning soaps. This is why formulation matters—two soaps can both be “real soap,” yet perform very differently.
A common concern people have when they see ingredients like sodium hydroxide or potassium hydroxide on a label is that these are harsh or dangerous chemicals. While that is true in their raw form, properly formulated soap does not contain free alkali. The reaction is carefully controlled so that the hydroxide is fully consumed during saponification, leaving behind the finished soap and naturally occurring glycerin.5
Finished soaps are typically alkaline, with a pH in the range of about 9 to 10. This is a natural result of the chemistry of fatty acid salts, and not an indication that unreacted lye remains in the product.
Despite being an ancient process, soapmaking is still widely used today because of its simplicity, transparency, and reliance on well-understood chemistry. When you see ingredients like “sodium cocoate,” “sodium olivate,” or “potassium oleate” on a label, you are looking at the direct result of this saponification process.
Addressing Soap’s Limitations
One of the primary challenges with traditional soap is its interaction with hard water. Minerals such as calcium and magnesium can react with soap to form insoluble residues, commonly referred to as “soap scum.”4 This can leave buildup on surfaces, fabrics, and even the skin.
However, this limitation is often overstated. Soap performance can be significantly improved through thoughtful formulation. For example, water softening agents such as sodium carbonate can help reduce mineral interactions, and the selection of specific fatty acids can influence how readily soap forms residues.
In other words, soap scum is not an unavoidable flaw—it is, to a large extent, a formulation challenge. And like most formulation challenges, it can be mitigated when properly understood.
That said, the inconvenience of soap scum in hard water environments was one of the key drivers behind the development and widespread adoption of synthetic detergents, which are designed to remain effective regardless of water hardness.
How Are Detergents Made?
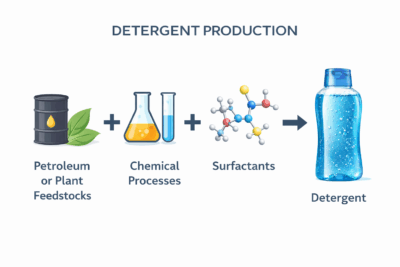
Unlike traditional soap, detergents are not produced through saponification. Instead, they are manufactured through a series of chemical processes that create synthetic surfactants designed for specific performance characteristics.7,8
Most modern detergents begin with raw materials derived from petroleum or plant-based feedstocks, which are chemically modified to produce surfactant molecules. These molecules are engineered to reduce surface tension, allowing water to more effectively interact with oils, dirt, and other residues.7
Detergents are typically classified based on the electrical charge of their surfactant molecules:
- Anionic surfactants (such as sodium lauryl sulfate or linear alkylbenzene sulfonates) are widely used for their strong cleansing and foaming properties.
- Nonionic surfactants (such as alcohol ethoxylates) are valued for their ability to remove oils and function well in hard water.
- Amphoteric surfactants (such as cocamidopropyl betaine) are often included to improve mildness and reduce irritation.
One of the key advantages of detergents is that they are less likely than soap to react with minerals in hard water, which allows them to maintain performance without forming insoluble residues.8
However, this increased formulation flexibility comes with added complexity. Many detergent surfactants are produced through multi-step chemical processes such as sulfonation, sulfation, or ethoxylation. While these processes are designed to yield stable and effective ingredients, they can also introduce the potential for residual byproducts or impurities if not properly controlled and purified.
As a result, modern detergent formulations often involve not just surfactants, but also stabilizers, preservatives, and other functional additives. This complexity makes ingredient transparency—and the quality control practices of the manufacturer—especially important when evaluating these products.
Contaminants, Toxicology, and Safety Concerns
When comparing soaps and detergents, most discussions focus only on cleaning performance. But an equally important—and often overlooked—consideration is toxicology.
With traditional soap, the chemistry is relatively simple and transparent. The starting materials (fats or oils and an alkali) are well understood, and the end product consists primarily of fatty acid salts and glycerin. In contrast, modern detergent formulations can involve multiple synthetic surfactants, stabilizers, preservatives, and processing aids—many of which are not fully disclosed to the consumer.
This creates two layers of uncertainty. First, the origin of the raw materials is often unclear. While some detergents may be derived from plant-based feedstocks, many are produced using petrochemical intermediates that undergo extensive chemical modification.7 Second, and more importantly, the final product is rarely a single compound. It is a mixture of multiple ingredients, along with trace impurities or byproducts that may remain from the manufacturing process.
One well-documented example is 1,4-dioxane, a compound classified as a probable human carcinogen.9 It is not intentionally added to products, but can form as a byproduct during a process known as ethoxylation, which is used to manufacture certain types of nonionic surfactants.10 Multiple analyses have detected 1,4-dioxane in consumer cleaning and personal care products, sometimes at measurable levels.10
Similarly, formaldehyde and formaldehyde-releasing compounds may be present in some formulations, either as preservatives or as breakdown products. Formaldehyde is a known human carcinogen and a well-established cause of contact dermatitis in sensitive individuals.11,12
In addition to these concerns, certain surfactants themselves have been associated with skin irritation or allergic reactions. For example, cocamidopropyl betaine has been linked to contact dermatitis, often due to impurities introduced during manufacturing rather than the compound itself.13
None of this means that all detergent-based products are inherently unsafe. However, it does highlight an important point: the safety of a product cannot be assumed based solely on its intended function. It depends on the full formulation, the manufacturing process, and the level of transparency provided by the manufacturer.
When ingredient lists are incomplete or generalized—using terms like “anionic surfactants” or “fragrance”—it becomes difficult for consumers to evaluate what they are actually being exposed to. And in many cases, it is not just a single ingredient that matters, but the cumulative effect of repeated exposure to complex chemical mixtures over time.
So What Is Better—Soap or Detergent?
At this point, the question isn’t simply which one cleans better. Both soaps and detergents are effective at removing dirt and oils. The more meaningful question is how much you know about what you’re using—and how much you’re willing to trust what you don’t.
Soap offers a level of simplicity that is difficult to ignore. The chemistry is straightforward, the ingredients are typically recognizable, and the end product is relatively easy to understand. That doesn’t automatically make every soap superior, but it does make it easier to evaluate.
Detergents, by comparison, are often more complex. They are engineered for performance, stability, and consistency, particularly in challenging conditions like hard water. But that performance can come with tradeoffs—more intricate formulations, less transparent ingredient disclosures, and a greater reliance on manufacturing controls to ensure purity and safety.
This doesn’t mean all detergents are problematic, nor does it mean all soaps are ideal. What it does mean is that the burden shifts to the consumer. If a product does not clearly disclose what is in it—or uses vague terms that obscure its composition—it becomes difficult to make an informed decision.
So the answer is not simply “soap” or “detergent.” The better choice is the one you can fully understand, evaluate, and trust. And in many cases, that begins with choosing products that prioritize transparency over marketing.

How to Tell If You’re Buying Soap or a Detergent
One of the biggest challenges for consumers is that many products labeled as “soap” are not actually soap. Fortunately, there are a few reliable ways to tell the difference—if you know what to look for.
Look for saponified oils. True soap is made from fats or oils that have been reacted with an alkali. On an ingredient label, this often appears as terms like “sodium cocoate,” “sodium olivate,” “sodium palmate,” or “potassium oleate.” These names indicate that the oils have undergone saponification and are now present as fatty acid salts—the defining characteristic of real soap.
Watch for synthetic surfactants. If you see ingredients such as sodium lauryl sulfate (SLS), sodium laureth sulfate (SLES), cocamidopropyl betaine, or similar compounds listed as primary ingredients, you are looking at a detergent-based cleanser, even if the product is marketed as “soap.”
Be cautious with vague labeling. Terms like “anionic surfactants,” “cleansing agents,” or simply “fragrance” do not provide meaningful insight into what is actually in the product. These generalized labels make it difficult to evaluate the formulation and may obscure important details about ingredient composition.
Understand that form matters. Liquid “soaps” are almost always detergent-based formulations. While true liquid soaps do exist (typically made with potassium hydroxide), they are far less common than synthetic alternatives due to cost, stability, and formulation challenges.
Marketing terms are not reliable indicators. Words like “natural,” “gentle,” or “plant-based” are not regulated in a way that guarantees a product is true soap. These terms are often used broadly and should not be relied upon without reviewing the ingredient list.
Ultimately, the ingredient list is your most reliable source of information. Once you know what to look for, the difference between soap and detergent becomes much easier to identify.
Looking Beyond the Marketing
One of the most effective ways to mislead is not to present something entirely false, but to present something partially true—just enough to shape perception. The discussion around soap versus detergents often follows this pattern.
Yes, soap has limitations. Soap scum is real, particularly in hard water conditions. But that limitation is frequently overstated, and more importantly, it is heavily influenced by formulation. With a proper understanding of the chemistry, many of these issues can be reduced or managed.
What is far less discussed is the toxicological side of the equation. The conversation is often framed around performance—how well something cleans, how it behaves in hard water, how stable it is on a shelf—while questions about long-term exposure, formulation complexity, and ingredient transparency receive far less attention.
Modern detergent systems can involve multiple surfactants and processing steps, some of which are known to introduce impurities if not carefully controlled. For example, ethoxylated ingredients can contain trace amounts of 1,4-dioxane as a byproduct of manufacturing, and certain preservatives or contaminants have been associated with skin irritation or sensitization.9–13 In some cases, it is not the primary ingredient, but the residuals or byproducts that raise concern.
There are also important nuances that are often lost in simplified discussions. For instance, while some surfactants are synthesized using petrochemical intermediates, the presence of a hazardous starting material does not necessarily mean it is present in the final product. However, it does reinforce the importance of manufacturing quality, purification, and transparency—factors that are rarely visible to the end user.
Ultimately, the debate between soap and detergent is often framed too narrowly. It is not just about chemistry or performance—it is about understanding what goes into a product, how it is made, and what you are repeatedly exposing yourself to over time.
Without that information, the comparison is incomplete.
And if you don’t know what’s in a product, you’re not really choosing it—you’re trusting it.
For Health,
Rob
3. Smulders E, von Rybinski W, Sung E, et al. Laundry Detergents. Wiley-VCH Verlag; 2007.
4. Bajpai D. Laundry detergents: an overview. J Oleo Sci. 2007;56(7):327–340.
5. Cavitch SM. The Soapmaker’s Companion. Storey Publishing; 1997.
6. Rosen MJ, Kunjappu JT. Surfactants and Interfacial Phenomena. 4th ed. Wiley; 2012.
7. Smulders E, von Rybinski W, Sung E, et al. Laundry Detergents. Wiley-VCH Verlag; 2007.
8. Bajpai D. Laundry detergents: an overview. J Oleo Sci. 2007;56(7):327–340.
9. International Agency for Research on Cancer (IARC). 1,4-Dioxane. Monographs on the Evaluation of Carcinogenic Risks to Humans.
10. U.S. Food and Drug Administration. 1,4-Dioxane in Cosmetics: A Manufacturing Byproduct. Updated 2023.
11. International Agency for Research on Cancer (IARC). Formaldehyde. Monographs on the Evaluation of Carcinogenic Risks to Humans.
12. Thyssen JP, et al. Contact dermatitis caused by formaldehyde and formaldehyde-releasers. Contact Dermatitis. 2010.
13. Fowler JF Jr. Cocamidopropyl betaine allergy and irritant reactions. Dermatitis. 2008.