
In response to the COVID-19 pandemic, many manufacturers had to act quickly to address widespread hand sanitizer shortages, as demand for basic ingredients surged. While many followed the World Health Organization (WHO) recommended formula, others did not. The U.S. Food and Drug Administration (FDA) reported cases of serious injury, including blindness and death, linked to hand sanitizers containing methanol or improperly formulated products.
This article was originally written and published July 2020 during the early stages of the COVID-19 pandemic when hand sanitizer shortages led to widespread formulation issues. It has been updated to reflect current understanding while preserving the original context.
In case you didn’t know, substances applied to the skin can be absorbed to varying degrees, and hand sanitizer is no exception. While many people are aware of this, not all types of alcohol used in these products carry the same safety profile.
When COVID first disrupted everyday life, demand for hand sanitizer and its ingredients increased dramatically. In my experience, sourcing those ingredients became extremely difficult, and pricing for basics like 99% isopropyl alcohol increased to levels that were, frankly, hard to believe. It wasn’t just alcohol—glycerin, food-grade hydrogen peroxide, and other components all became harder to find and more expensive.
With empty shelves and limited supply, many manufacturers moved quickly to try to meet demand. During this time, the FDA announced temporary policy changes to allow more facilities to produce hand sanitizer. These changes focused on increasing supply, not altering recommended formulations or ingredient safety expectations.
*From my own experience, we made multiple attempts to purchase bulk quantities of 99% isopropyl alcohol, only to have orders cancelled due to limited availability. Not long after, some of those same sources began offering finished hand sanitizer products instead. It was a frustrating experience, and a clear example of how quickly priorities can shift when demand spikes.
So what were manufacturers and re-sellers to do? Many moved quickly to meet demand. Unfortunately, during this time, some products entered the market that were improperly formulated or contaminated. The FDA reported cases of serious injury, including blindness and death, linked to hand sanitizers containing methanol.1
Methanol, sometimes referred to as wood alcohol, is still an alcohol, but a dangerous one. It is chemically similar to ethanol (drinking alcohol), but the body processes it very differently. Even relatively small amounts can be toxic. Methanol exposure has been associated with severe outcomes including damage to the optic nerve, which can result in permanent blindness, and in higher doses, death.1
To put that into perspective, very small volumes—on the order of milliliters—can be enough to cause serious harm. That is not a large amount. For comparison, a standard drink is typically around 1.5 ounces of distilled spirits. The difference is that ethanol is metabolized differently, while methanol breaks down into toxic byproducts that can damage the nervous system.
Unfortunately, there have been cases where individuals—both children and adults—ingested hand sanitizer under the assumption that it contained ethanol, only to later discover it contained methanol or was contaminated with it.1
The FDA identified numerous hand sanitizer products contaminated with methanol and issued warnings and recalls. In some cases, methanol was not listed on the ingredient label, making it difficult for consumers to distinguish between safe and unsafe products without additional testing.1
But just because you’re not drinking hand sanitizer doesn’t automatically mean there is no risk. Methanol can be absorbed through the skin to some extent, and the FDA has warned that individuals using contaminated products may be at risk for adverse effects.1
If you experience symptoms such as nausea, headache, blurred vision, dizziness, or seizures after using a hand sanitizer, it is important to seek medical attention as soon as possible to reduce the risk of serious or permanent harm.
So when choosing a hand sanitizer, remember that not all products are created equal. Reading the ingredient list is key. The WHO has provided clear guidance on hand sanitizer formulations, emphasizing simple, well-defined ingredient combinations without unnecessary additives.2
A typical formulation should be straightforward to recognize by turning the product over and reviewing the ingredients. It should clearly identify either ethyl alcohol or isopropyl alcohol as the active ingredient, along with components such as glycerin, hydrogen peroxide, and water. According to FDA labeling requirements, this information should appear in a standardized “Drug Facts” panel similar to the example shown below:
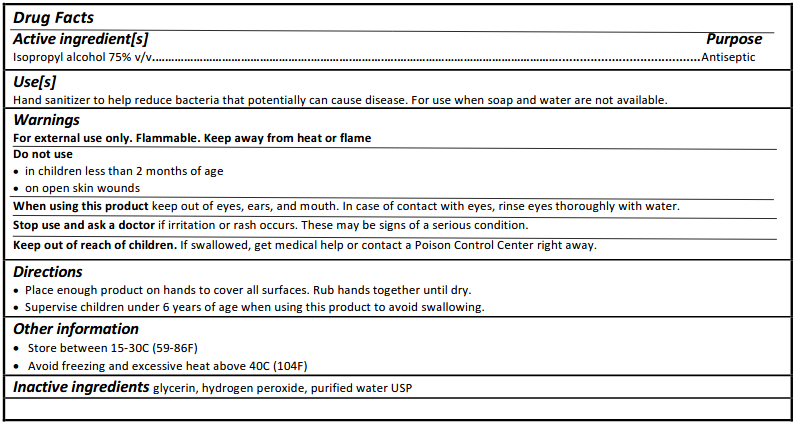
If the type of alcohol is not clearly identified on the label, it’s best to avoid the product or contact the manufacturer for clarification.
While ingestion of hand sanitizer is not its intended use, both ethyl alcohol and isopropyl alcohol can be harmful if swallowed. However, unlike methanol, they do not produce the same highly toxic metabolites associated with severe neurological damage.1
Consider the brand and whether you trust them. Not all manufacturers operate with the same priorities, and that matters. From my experience in this industry, finding suppliers and partners who consistently prioritize quality and transparency can be challenging, especially during times of disruption.
Over the years, and particularly during COVID, I found myself spending more time researching raw ingredient sources than ever before. Supply chains shifted, availability changed, and it became increasingly important to verify not just what was being sold, but where it was coming from and how it was being handled.
While I understand the challenges manufacturers face in sourcing materials during uncertain times, it also highlights the importance of transparency. When demand increases and supply is limited, it becomes even more important for consumers to pay attention to who they are buying from and to choose brands that clearly communicate what they are doing and why.
Consider making your own. We previously provided detailed instructions based on the WHO formulation using either isopropyl alcohol or ethyl alcohol. As availability of ingredients has improved, sourcing components has become more manageable.
Having these ingredients on hand can give you more control over what you are using. For those who prefer a do-it-yourself approach, this can be a practical way to ensure you know exactly what is going into your product. That said, proper formulation and handling are important, and any product, whether store-bought or homemade, should be used as directed.
And lastly, if you choose to drink… please don’t drink your hand sanitizer.

For health,
Tober
References:
1. U.S. Food and Drug Administration (FDA). FDA Updates on Hand Sanitizers with Methanol. Available at: https://www.fda.gov/drugs/drug-safety-and-availability/fda-updates-hand-sanitizers-methanol
2. World Health Organization (WHO). Guide to Local Production: WHO-recommended Handrub Formulations. Available at: https://www.who.int/publications/i/item/WHO-IER-PSP-2010.5
Nature's Complement is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program. If you purchase products on Amazon through any of our affiliate links, we get a small percentage of the transaction, at no extra cost to you. We spend a lot of time writing the articles on this site, and all this information is provided free of charge. When you use our affiliate links, you support the writing you enjoy without necessarily buying our products. (However we would appreciate if you would do that too!) Thank you for helping to support our work, however you choose to do so.
These statements have not been evaluated by the Food and Drug Administration. This information and/or products are not intended to diagnose, treat, cure or prevent any disease.


