
Public controversy surrounding major advertising campaigns from large consumer product companies has, at times, led to calls for boycotts. While much of that attention tends to focus on messaging and social issues, it often overlooks a far more consequential concern: the ingredients used in everyday personal care and household products. Rather than weighing in on advertising decisions, this article redirects that attention toward the growing body of scientific evidence surrounding certain chemical exposures—and what they may mean for human health. Read on to better understand these concerns and how to make more informed choices for yourself and your family.
If you are not familiar with the background of this topic, numerous mainstream outlets have covered the public reaction to a widely discussed advertising campaign that aimed to address social behaviors such as bullying and harassment. The response was mixed, with some viewers supporting the message and others criticizing its tone and interpretation.
Rather than focusing on the advertisement itself, it is worth stepping back and considering a broader question: how closely do the standards promoted in public messaging align with the standards applied to product formulation and consumer safety? While messaging can spark important conversations, it should not overshadow ongoing scientific concerns about the ingredients used in many everyday products. A growing body of research has raised questions about whether certain commonly used chemicals may carry health risks for both men and women, with particular concern for developmental and reproductive effects in males. This is the issue we will examine more closely.
A growing body of scientific research has raised concerns that certain chemical ingredients commonly used in personal care and household products may interfere with normal hormonal function.1–3 These substances—often referred to as endocrine-disrupting chemicals (EDCs)—include compounds such as phthalates, parabens, triclosan, certain glycol ethers, and synthetic colorants like tartrazine.2,4–6 Studies in both animal models and human populations have shown that some of these chemicals can interact with hormone signaling pathways, including those involved in male reproductive development.1,3,7
Of particular concern is exposure during critical windows of development, such as fetal growth, infancy, and early childhood, where hormonal signaling plays a central role in shaping long-term physiology.1,2,8
Research has associated certain exposures with changes in reproductive markers, altered hormone levels, and developmental differences in males.3,7,9 While the exact magnitude and implications of these effects continue to be studied, the consistency of findings across multiple lines of evidence raises important questions about long-term health impacts and warrants careful consideration.
Section 1:
Biological Effects on Male Development and Function
To begin, it is important to clearly define what is meant in a biological context when discussing changes to male development. In medical and scientific literature, alterations in male reproductive development and physiology may include measurable changes such as reduced sperm counts, decreased levels of testosterone and dihydrotestosterone (DHT), differences in reproductive anatomy (including conditions such as hypospadias and cryptorchidism), and shifts in secondary sex characteristics.10–12 Additional observations in research settings have included changes in muscle and bone mass, metabolic differences, and the development of conditions such as gynecomastia.10,11
These outcomes are typically evaluated within the framework of endocrine function, particularly the role of androgens—hormones such as testosterone and DHT produced primarily by the testes. Androgens play a central role in regulating gene expression involved in male development, especially during critical periods such as fetal growth, infancy, and puberty.11,12 Disruptions to these hormonal signaling pathways—whether through reduced androgen activity or interference at the receptor level—can lead to measurable changes in developmental and physiological outcomes. A growing body of research has demonstrated that certain environmental and chemical exposures have the potential to interfere with these processes, as will be explored in the sections that follow.1,3,10
 Testosterone, the molecular version.
Testosterone, the molecular version.
So what mechanisms may underlie these observed changes? A significant area of scientific investigation has focused on the previously mentioned chemicals known as endocrine-disrupting chemicals (EDCs), including a subset often referred to as xenoestrogens—compounds that can mimic or interfere with the activity of endogenous hormones such as estrogen.1,2,13 The endocrine system regulates hormone production, signaling, and balance throughout the body, and even small disruptions during critical developmental periods can have measurable biological effects.
Some of these compounds have been shown to bind to estrogen receptors or otherwise influence hormone signaling pathways in both males and females.1,13In experimental models, this can lead to altered gene expression and shifts in hormonal balance, including interactions with androgen pathways responsible for male development.3,12 Additionally, normal endocrine function relies on tightly regulated feedback mechanisms that maintain hormonal equilibrium. Interference with these systems—particularly during sensitive stages such as fetal development—may contribute to changes in how these pathways are regulated over time. While the precise effects depend on factors such as dose, timing, and individual susceptibility, these mechanisms provide a biologically plausible explanation for the associations observed in reproductive and developmental research.
 Estradiol.
Estradiol.
It is important to recognize that both males and females naturally produce and rely on a balance of hormones, including small amounts of estrogen and testosterone. These hormones play essential roles in normal physiology. However, increasing attention has been given to whether modern environmental exposures may be altering this balance. In particular, certain endocrine-disrupting chemicals have been shown in experimental and epidemiological studies to interact with estrogen receptors and influence hormonal signaling pathways.1,3,13
One of the most frequently cited indicators of potential changes in male reproductive health is the observed decline in sperm counts over time. In 2017, a large meta-analysis conducted by researchers from Hebrew University and the Icahn School of Medicine at Mount Sinai analyzed data from 185 studies involving nearly 43,000 men across North America, Europe, Australia, and New Zealand.14 The study reported a significant decline in sperm concentration and total sperm count over a period of approximately four decades, with reductions exceeding 50% in some populations.14 While the causes of this trend are likely multifactorial—including lifestyle, environmental, and chemical exposures—the findings have raised substantial concern within the scientific community and have prompted ongoing investigation into the role of environmental factors in male reproductive health.

These findings are not isolated. Additional research and reviews from major public health and scientific bodies, including the National Institutes of Health and the Centers for Disease Control and Prevention, have examined trends in male reproductive health and the potential role of environmental and lifestyle factors.3,10,12 Declining sperm counts are only one aspect of a broader pattern that has been investigated over the past several decades.
Epidemiological studies have explored changes in the incidence of conditions such as hypospadias and cryptorchidism, as well as shifts in testosterone levels in adult males.11,12,16 Some analyses suggest that these conditions may be influenced by a combination of genetic, environmental, and developmental factors, with particular attention given to exposures occurring during early life.3,12 While the data are complex and not always uniform across regions and study designs, the overall body of evidence has led many researchers to examine whether environmental exposures—including endocrine-disrupting chemicals—may contribute to observed changes in male reproductive health over time.
Taken together, these findings highlight the importance of continued investigation into environmental influences on human development and underscore the need for greater awareness of potential risk factors.
Section 2:
Additional Health Considerations Related to Endocrine Disruption
While much of the discussion surrounding endocrine-disrupting chemicals has focused on male reproductive development, it is important to recognize that these compounds have been studied in relation to a broader range of potential health effects. Research has explored associations between certain endocrine-disrupting exposures and multiple physiological systems, reflecting the widespread role that hormones play throughout the body.1–3,13
These potential effects are not limited to one population or outcome. Instead, they span developmental, metabolic, and reproductive processes, and may vary depending on timing, duration, and level of exposure. The following sections highlight several areas that have been examined in the scientific literature.

Early Puberty in Girls
A number of studies have investigated the potential relationship between endocrine-disrupting chemicals and the timing of pubertal development in girls.17–19 Some research suggests that exposure to estrogenic compounds may be associated with earlier onset of certain developmental markers, including breast tissue development.17,18 One area of interest is isolated premature thelarche (IPT), a condition characterized by early breast development without other signs of puberty. In smaller clinical studies, differences in estrogenic activity have been observed between groups with varying levels of exposure to endocrine-disrupting compounds.18
These findings are supported by broader research examining how hormonally active compounds may influence endocrine signaling during sensitive developmental periods.1,2,13 While individual studies vary in size and design, the overall body of evidence has led researchers to further explore whether environmental exposures may play a role in shifts in pubertal timing. Importantly, early onset of puberty has been identified as a potential risk factor for certain long-term health outcomes, including increased risk of breast cancer later in life.19
Additional research has examined compounds such as Bisphenol A (BPA), which is widely used in plastics, food packaging, and thermal receipt paper. Some studies have investigated whether BPA exposure may be associated with precocious puberty, although findings remain an area of ongoing research.20 While BPA is not typically used in personal care products, its widespread presence in the environment highlights the broader context of cumulative chemical exposure.
Given the importance of hormonal signaling during development, many researchers emphasize the value of minimizing unnecessary exposures—particularly during early life stages—where feasible and practical.
Breast Cancer in Women
Early developmental factors and lifetime hormonal exposure have long been recognized as important considerations in breast cancer risk. Research has shown that cumulative exposure to estrogens—both endogenous and exogenous—can influence the development and progression of hormone-sensitive breast cancers.19,21 This has led scientists to examine whether certain endocrine-disrupting chemicals, particularly those with estrogen-like activity, may contribute to these pathways.
Experimental studies have provided insight into how some of these compounds may interact with breast tissue at the cellular level. In vitro research has demonstrated that certain xenoestrogenic compounds, including phthalates, bisphenol A (BPA), and parabens, can influence estrogen receptor activity and gene expression in human breast epithelial or breast cancer cells.22–24 These findings do not directly translate to clinical outcomes on their own, but they do provide biologically plausible mechanisms that continue to be investigated in broader epidemiological and clinical research.
Taken together, the available evidence highlights the importance of understanding how cumulative exposures may interact with hormone-sensitive tissues over time. While research in this area is ongoing and continues to evolve, these findings have contributed to increased scientific interest in the role of environmental factors in breast cancer risk and progression.

A common argument in the safety assessment of many chemical ingredients is that individual exposures occur at levels considered too low to produce biological effects. However, an important area of research has examined whether multiple low-dose exposures—across diet, household products, and personal care items—may combine to produce measurable hormonal activity.22,25 Some studies have demonstrated that mixtures of compounds with estrogenic properties can produce additive or even synergistic effects, even when each individual component is present at a low concentration.25
This concept is particularly relevant when considering real-world exposure patterns. Surveys have shown that individuals may use numerous personal care products daily, each containing multiple ingredients.26 As a result, cumulative exposure to hormonally active compounds can occur across multiple sources throughout the day.

Additionally, biomonitoring studies have detected various endocrine-disrupting chemicals or their metabolites in human tissues and fluids, indicating that exposure and absorption do occur.1,8 One of the ongoing challenges in toxicology is that traditional safety assessments often evaluate substances individually, whereas real-world exposure involves complex mixtures. This has led to increasing scientific interest in understanding cumulative exposure and potential interactions between compounds, which remain areas of active research.
Refocusing on breast cancer, a number of studies have evaluated the potential role of certain chemical exposures in influencing disease progression. Some research has examined parabens specifically, noting their ability to interact with estrogen receptors and influence cellular behavior in breast tissue models.23,27
Additional in vitro studies have reported that parabens can affect not only cellular proliferation, but also migratory and invasive characteristics of human breast cancer cells.27 While findings from cell-based models do not directly translate to clinical outcomes, they contribute to an understanding of possible biological mechanisms and help guide further investigation.
Epidemiological research has also explored patterns of product use and potential differences in exposure. Some studies have examined whether higher usage of personal care products may contribute to differences in cumulative exposure to hormonally active compounds, and how this may relate to observed disparities in breast cancer risk.28 These findings remain an area of ongoing research and should be interpreted within the broader context of multiple contributing factors.
Other exposures, including certain compounds found in hair dyes, have also been investigated for possible associations with breast cancer risk, although results vary depending on study design and population.29
Given the complexity of breast cancer risk factors and the role of hormonal signaling, many researchers emphasize the importance of understanding and, where practical, minimizing unnecessary exposures to hormonally active compounds.
 A light blue ribbon is the symbol for prostate health month.
A light blue ribbon is the symbol for prostate health month.
Benign Prostatic Hyperplasia (BPH) and Prostate Cancer in Men
Concerns related to endocrine-disrupting chemicals are not limited to female health. A growing body of research has examined whether hormonally active compounds may also influence prostate health in men.30–32 Review articles drawing on epidemiological and experimental data have explored how certain exposures—including compounds such as bisphenol A (BPA) and other endocrine-disrupting chemicals—may interact with hormonal pathways involved in prostate development and disease progression.30,31
Benign prostatic hyperplasia (BPH) is commonly observed in men as they age, but this does not mean that aging itself is the underlying cause. Rather, BPH is increasingly understood as a condition influenced by changes in hormonal signaling over time, particularly the balance between androgens and estrogens within prostate tissue.30,31 The frequent identification of BPH later in life may reflect the cumulative effects of these biological and environmental influences, rather than age alone as a causal factor.
Some research has examined the role of estrogen signaling in prostate tissue, including the effects of both endogenous hormones and external exposures. In clinical contexts, modulation of hormonal pathways—including anti-estrogen approaches—has been explored as part of prostate cancer management, further underscoring the importance of hormonal regulation in this tissue.30
Although the precise contribution of environmental exposures remains an area of ongoing research, these findings highlight the importance of considering how cumulative hormonal influences—over time—may affect prostate health.
Research has highlighted that the prostate may be particularly sensitive to hormonal influences during critical developmental windows. Studies have noted that exposure to estrogenic compounds during early life—especially during fetal development—can influence how prostate tissue develops and responds later in life.30,31,33
Experimental research has also explored how estrogens may contribute to prostate carcinogenesis within androgen-supported environments, pointing to complex interactions between hormonal pathways in prostate disease.34 Additional reviews have examined the role of estrogen receptors in prostate growth, differentiation, and disease states, including prostatitis, benign prostatic hyperplasia (BPH), and prostate cancer.30
Animal studies have further investigated the potential effects of environmental exposures. For example, some research has found that low-dose exposure to compounds such as bisphenol A (BPA) may influence prostate cell proliferation and interact with androgen-driven processes in experimental models.31 Other studies have shown that fetal exposure to certain estrogenic compounds can alter prostate development and result in measurable changes in prostate size later in life in animal models.33
These findings highlight the importance of developmental timing in endocrine signaling. While results from animal and experimental studies do not directly translate to human outcomes, they provide insight into mechanisms by which early-life exposures may influence long-term health. As a result, many researchers emphasize the importance of minimizing unnecessary exposures during sensitive developmental periods, including pregnancy, where feasible and practical.

Finally, some research has explored how endocrine-disrupting chemicals may influence prostate biology at the cellular level, including effects on stem cell populations that play a role in tissue development and regeneration.35 These findings suggest that certain exposures may alter how prostate cells respond to hormonal signals over time, which could have implications for disease susceptibility.
Endocrine-disrupting compounds are widespread in the environment, and multiple pathways of exposure have been identified, including diet, household products, and personal care items.1,8 As discussed throughout this article, particular attention has been given to exposures occurring during early developmental stages, where hormonal signaling plays a critical role in long-term physiological outcomes.
While there is a substantial and growing body of research examining the role of endocrine-disrupting chemicals in both prostate and breast health, the focus of this article remains on male developmental and reproductive considerations, along with practical steps individuals can take to better understand and manage their exposures.

Section 3
Ingredients of Concern and How to Approach Them
With an understanding of the underlying biology, the next question becomes: which types of ingredients are commonly discussed in the context of endocrine disruption, and how can individuals make more informed choices? A number of chemical compounds used in personal care and household products have been studied for their potential to interact with hormonal systems, particularly those with estrogenic or endocrine-disrupting properties.1–3,13
It is important to note that the scientific literature in this area is extensive and continues to evolve. The ingredients highlighted below are among those that have been frequently examined in peer-reviewed research for their potential biological activity. While not exhaustive, they represent commonly encountered compounds that have been investigated for their interactions with endocrine pathways and their possible effects on human health.
These considerations may be particularly relevant during sensitive developmental periods, including pregnancy, infancy, and early childhood, when hormonal signaling plays a critical role in growth and development.1,2 As with many areas of environmental health, the goal is not to eliminate all exposures—which is often impractical—but to better understand where exposures occur and reduce unnecessary or avoidable sources where feasible.
The following sections provide an overview of selected ingredients that have been studied for their endocrine activity, along with references to the scientific literature supporting these investigations.
 Generic phthalate structure.
Generic phthalate structure.
1. Phthalates – Often Associated with “Fragrance” or “Parfum”
Phthalates are a class of chemicals widely used in consumer products, including certain personal care items such as nail polish, hair products, and fragranced formulations.4,7 In many cases, phthalates are not listed individually on product labels. Instead, they may be included as part of ingredients described generically as “fragrance,” “parfum,” or “perfume,” due to regulatory allowances that permit proprietary fragrance formulations to be protected as trade secrets.4
This labeling practice can make it difficult for consumers to determine whether a product contains phthalates or other fragrance-related compounds. Research has shown that some phthalates can interact with hormonal systems, particularly by influencing androgen signaling pathways involved in male reproductive development.4,7
Because fragranced products are widely used and often applied daily, they can represent a consistent and repeated source of exposure for many individuals. This is especially relevant when “fragrance” or “parfum” is used as a catch-all term, as it may include multiple undisclosed components. For this reason, some consumers choose to limit or avoid products containing undisclosed fragrance ingredients, or instead select products that fully disclose their contents or are labeled as phthalate-free.

Independent investigations have attempted to better characterize the composition of fragranced consumer products by using analytical techniques such as gas chromatography–mass spectrometry (GC-MS). These analyses have identified the presence of phthalates and other compounds in products where such ingredients were not individually disclosed on labels, but instead included under general terms such as “fragrance.”4,36
Findings from these types of studies highlight the challenges associated with ingredient transparency in fragranced formulations and have contributed to ongoing discussions about labeling practices and consumer awareness.

Analyses of fragranced products have also highlighted the complexity of what may be included under the single term “fragrance.” Regulatory frameworks in the United States allow manufacturers to group multiple chemical components under this label to protect proprietary formulations. As a result, a single “fragrance” ingredient may represent a mixture of numerous individual compounds rather than a single substance.4
This labeling convention can make it challenging for consumers to fully understand the composition of fragranced products. While the term “fragrance” often suggests a single scent-related ingredient, it may instead refer to a combination of compounds with varying functions, including scent enhancement, stabilization, and longevity.
Phthalates are one class of compounds that have been used in some fragranced formulations to help stabilize or prolong scent. Other components, such as synthetic musks, may also be present in certain products. These compounds have been studied for their potential biological activity, including interactions with hormonal pathways, although effects can vary depending on the specific chemical, dose, and context of exposure.4,6
Trace levels of certain fragrance-related compounds, including synthetic musks, have been detected in environmental water sources, reflecting the widespread use and persistence of these chemicals.37 This highlights how everyday product use can contribute to broader environmental exposure pathways, although concentrations and health implications can vary depending on multiple factors.
Returning to phthalates, these compounds have been the subject of extensive research due to their widespread use as plasticizers and fragrance stabilizers. Studies have examined their presence in a variety of consumer products, as well as potential routes of human exposure, including dermal absorption from personal care items.4,36,38 Regulatory and scientific assessments, including those from agencies such as Health Canada, have evaluated exposure levels and potential health implications.
A substantial body of research has investigated the effects of phthalate exposure on reproductive health, particularly in males. Findings from both human and animal studies have reported associations with changes in hormone levels and markers of reproductive development.4,7 While results vary across studies, the consistency of these observations has contributed to ongoing scientific and regulatory interest in these compounds.
Experimental studies have investigated the effects of phthalate exposure on male reproductive development, including early research demonstrating testicular changes in animal models following exposure to certain phthalate esters.39 These effects were often more pronounced when exposure occurred during early developmental stages, rather than in fully mature animals, highlighting the importance of timing.
One way scientists measure male development in both animals and humans is by looking at something called anogenital distance (AGD), which is simply the distance between the anus and the genitals. This measurement tends to be longer in males and shorter in females, and it reflects how much male hormone signaling occurred during development in the womb. In animal studies, prenatal exposure to certain phthalates has been associated with a shorter AGD in male offspring.7
Importantly, similar findings have been observed in human studies. Researchers have found that higher levels of phthalate exposure during pregnancy are associated with shorter AGD in male infants.40 This suggests that these exposures may influence aspects of male reproductive development before birth. While this type of research shows association rather than direct causation, the consistency between animal and human findings has raised significant scientific interest in this area.

Follow-up research expanded on these findings by examining additional developmental markers in male infants. In this work, researchers evaluated not only anogenital distance (AGD), but also related measures such as incomplete testicular descent, and introduced the use of the anogenital index (AGI)—which adjusts AGD relative to body size—to improve accuracy.40,41 These studies reported associations between higher prenatal phthalate exposure and changes in these developmental markers, further supporting the observations from earlier research.
The lead researcher on these studies, Shanna Swan, has contributed extensively to this field of research. As with many areas of emerging science, her work has been both influential and subject to scientific debate. Some critiques have focused on aspects of study design and statistical methods, while others have emphasized the need for replication and continued investigation. Despite differing interpretations, these studies have played a significant role in advancing research into how environmental exposures during pregnancy may influence early developmental outcomes.
When credible research raises concerns about the safety of widely used chemical ingredients, the appropriate response should be careful review, transparency, and a willingness to reevaluate product formulation where warranted. However, most consumers are not regularly exposed to scientific literature or emerging research findings, and many reasonably assume that products on the market have already been thoroughly evaluated for long-term safety.
In practice, change often moves slowly. Scientific findings may take years to be fully evaluated, debated, and incorporated into regulatory standards. During that time, products typically remain on the market, and ingredient transparency may still be limited. As a result, individuals are often placed in the position of needing to interpret available information and make personal decisions about which exposures they are comfortable accepting.

One important question raised by this research is the level of exposure at which measurable effects may occur. A study examining estimated daily phthalate exposures in mothers of male infants with reduced anogenital distance (AGD) explored this issue by analyzing multiple exposure pathways, including dermal contact, ingestion, and inhalation.42 The researchers noted that phthalates are rapidly metabolized and cleared from the body, yet the widespread presence of these compounds resulted in relatively consistent, ongoing exposure among study participants.
Notably, the estimated exposure levels associated with these developmental findings were substantially lower than existing regulatory reference doses (RfDs) established by the U.S. Environmental Protection Agency (EPA).42 This raises a meaningful concern: under certain conditions—particularly during sensitive developmental periods—biological effects may occur at exposure levels below those traditionally considered acceptable.
These findings do not establish precise thresholds for harm, but they do highlight a potential gap between regulatory assumptions and real-world exposure patterns. In particular, continuous low-dose exposures and cumulative effects may not be fully captured by conventional risk models, warranting continued investigation and cautious interpretation of existing safety limits. In practical terms, these findings indicate that measurable developmental effects have been observed at exposure levels significantly lower than current regulatory reference doses (RfDs).42 This raises a clear concern that existing safety thresholds may not fully account for sensitive developmental periods or continuous, real-world exposure patterns.
It is also worth noting that many of the foundational studies in this area were published in the early 2000s. As a result, the potential risks associated with phthalate exposure have been recognized in the scientific literature for decades. Despite this, questions remain regarding how effectively this body of evidence has been translated into updated formulations, regulatory standards, or consumer-facing transparency.

Additional research has examined early-life exposure beyond pregnancy, including the potential transfer of certain phthalates through breast milk. Some studies have reported associations between early postnatal exposure and measurable hormonal or developmental changes in infants, further emphasizing that exposure windows may extend beyond birth.43
Review papers summarizing both animal and human research have described a range of findings associated with perinatal phthalate exposure, including alterations in testicular development, changes in hormone levels, reduced anogenital distance (AGD), and other markers of male reproductive development.3,40,44 These observations, reported across multiple study types, have contributed to ongoing concern and continued investigation into early-life exposure effects.
A substantial number of additional studies—many dating back several decades—have examined the effects of phthalate exposure in animal models, particularly in rodents. These studies have consistently reported changes in testicular structure, sperm morphology, and overall reproductive function following exposure to various phthalate compounds.39,44–47 Collectively, this body of research has contributed significantly to the scientific understanding of how these chemicals may interact with male reproductive systems.
While animal studies are not interpreted in isolation, they provide important mechanistic insight and help inform the design and interpretation of human epidemiological research. The consistency of findings across multiple studies and time periods has been a key factor in driving continued investigation into potential human health implications.
Taken together, the available evidence suggests that routine exposure to certain phthalates—particularly through commonly used fragranced products—may represent a meaningful source of endocrine-active compounds in daily life. As a result, increasing attention needs to be placed on ingredient transparency and informed consumer choice when selecting personal care products.
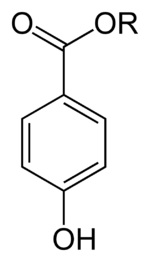 Paraben.
Paraben.
2. Parabens
Parabens are a class of preservatives that have historically been widely used in personal care products to prevent microbial growth and extend shelf life.5,23 In response to increased consumer awareness, many manufacturers have reduced or removed certain parabens from their formulations. However, parabens are still present in a number of products, and their use has not been eliminated.
In some cases, products may use blended preservative systems or alternative compounds that are less familiar to consumers. While these are not always parabens themselves, the increasing complexity of ingredient lists can make it more difficult to quickly identify which types of preservatives are being used, and if they include parabens under a different name. As a result, careful review of ingredient labels remains important.
Scientific studies have examined the ability of parabens to interact with estrogen receptors and influence hormone-related pathways.5,23 Although their activity is generally weaker than naturally occurring estrogens, their widespread use and potential for cumulative exposure have led to continued research and discussion regarding their long-term effects.
For consumers seeking to limit exposure, looking specifically for clearly labeled “paraben-free” products or those that fully disclose their preservative systems can help support more informed decision-making.
Several studies have examined the effects of specific parabens on male reproductive health. For example, research has reported decreases in sperm production associated with exposure to propylparaben, and other studies have found that butylparaben can adversely affect the male reproductive system at doses below established acceptable daily intake (ADI) levels.23,48 These compounds are not limited to personal care products—they have also been detected in certain food products, meaning exposure can occur through multiple pathways.
There are several common forms of parabens, including methylparaben, ethylparaben, propylparaben, and butylparaben. Studies have shown that these compounds can exhibit estrogen-like (xenoestrogenic) activity in both laboratory (in vitro) studies and animal models.5,23
Parabens have been widely used in skin care products for their antibacterial and preservative properties. However, animal studies—particularly involving butylparaben—have reported developmental effects in male offspring, including reduced anogenital distance, smaller testes, lower testosterone levels, and decreased sperm counts.49 These findings are notable because they mirror patterns observed with other endocrine-disrupting compounds.
Parabens have also been associated with changes in skin appearance in some formulations. Research suggests that these effects may be linked, at least in part, to their ability to interact with estrogen-related pathways, rather than providing purely structural or restorative benefits to the skin.
Research has also explored potential mechanisms by which parabens may affect male reproductive health. One review suggests that parabens may interfere with mitochondrial function in testicular cells. Mitochondria are often described as the “power plants” of cells, and disruption of their function can impair energy production and normal cellular activity. The authors propose that this interaction may help explain observed decreases in reproductive potential associated with paraben exposure.23
Additional studies have examined how parabens interact with multiple hormonal systems. Structure–activity research has shown that parabens can activate glucocorticoid receptors—hormone pathways involved in metabolism, inflammation, and immune regulation—indicating that their effects are not limited to estrogen signaling alone.50 This same line of research has also identified anti-androgenic activity in other commonly used compounds, such as triclosan, which will be discussed separately.
Further experimental work has demonstrated that parabens, along with certain other commonly used cosmetic ingredients such as homosalate, exhibit estrogenic activity across multiple cell types.24,51 Taken together, these findings reinforce an important point: parabens are biologically active compounds capable of interacting with several hormonal pathways.
While no single study provides a complete picture, the consistency of findings across mechanistic, animal, and cellular research supports the conclusion that parabens can influence hormone-related biological processes. This is particularly relevant when considering repeated, long-term exposure from everyday consumer products.

Triclosan.
3. Triclosan
Triclosan is an antimicrobial compound that was widely used for many years in antibacterial soaps and other personal care products. For a long time, it remained on the market under the assumption that it was both safe and effective. However, regulatory review eventually led the U.S. Food and Drug Administration (FDA) to determine that triclosan does not meet the standard of being Generally Recognized as Safe and Effective (GRASE) for use in over-the-counter (OTC) consumer antiseptic products.52
As a result, triclosan was removed from many antibacterial soaps, not because it demonstrated clear clinical benefit over standard soap and water, but because manufacturers were unable to provide sufficient evidence supporting both its safety and effectiveness for long-term use.52 This distinction is important—absence of demonstrated safety is not the same as confirmed safety.
Although its use has declined, triclosan has not been completely eliminated and may still be found in certain products or remaining inventory. For this reason, it remains relevant when evaluating ingredient lists.
From a biological standpoint, triclosan has been studied for its potential to interfere with hormone signaling pathways, including anti-androgenic effects observed in experimental models.50 Given its widespread historical use and evidence of biological activity, triclosan serves as a clear example of how commonly used ingredients can remain in circulation long after questions about their safety have been raised.
For practical purposes, standard soap and water remain sufficient for routine hygiene, without the need for additional antimicrobial agents in most everyday settings.
An important takeaway from these examples is that changes in product formulation and regulatory guidance often occur gradually, and typically only after sufficient evidence accumulates. In some cases, concerns about certain ingredients are raised and debated in the scientific literature for years before widespread changes are implemented. As a result, there can be a gap between emerging research findings and the products currently available to consumers. This makes independent research and ingredient awareness an important part of making informed decisions.
As discussed earlier, triclosan has been studied for its ability to interfere with androgen signaling, including blocking activity at the androgen receptor and affecting pathways involved in testosterone production.50,53 Research has also reported changes in hormone-related regulators such as luteinizing hormone (LH) and follicle-stimulating hormone (FSH), both of which play key roles in male reproductive function.
Experimental (in vitro) studies have further shown that triclosan can influence gene expression at hormone receptors and may stimulate the growth of certain breast cancer cells under specific conditions.54 Other studies have reported that triclosan can interact with both androgen and estrogen receptors, indicating that its biological effects are not limited to a single hormonal pathway.50
Beyond reproductive hormones, triclosan has also been investigated for potential effects on thyroid function. Epidemiological studies have reported associations between higher triclosan exposure and altered thyroid hormone levels, suggesting broader endocrine system involvement.55
Although individual findings vary, review papers evaluating the overall body of evidence have concluded that triclosan demonstrates endocrine-disrupting activity across multiple systems. While the precise health implications continue to be studied, the consistency of these observations supports ongoing concern regarding its biological effects.
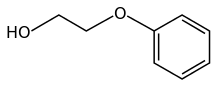 Phenoxyethanol, a glycol ether.
Phenoxyethanol, a glycol ether.
4+. Glycol Ethers (including ethylene glycol, phenoxyethanol, propylene glycol, and related compounds)
Glycol ethers represent a broad class of chemicals with a wide range of uses, from industrial applications such as antifreeze to roles in consumer products as solvents, preservatives, and stabilizers. Because this category includes many different compounds, their biological effects can vary significantly depending on the specific chemical and level of exposure.
Propylene glycol, in particular, is often cited as safe due to its classification as “Generally Recognized As Safe” (GRAS) by the U.S. Food and Drug Administration (FDA). However, GRAS status reflects specific use conditions and available data at the time of evaluation—it is not a blanket guarantee of safety under all exposure scenarios. Experimental studies have reported that high levels of exposure to propylene glycol can produce measurable physiological effects in animal models, including changes in liver weight and central nervous system depression.56,57
While propylene glycol is generally considered lower risk relative to some other glycol ethers, other compounds within this class have raised greater concern. Certain glycol ethers have been associated in research studies with reproductive and developmental effects, particularly in occupational or high-exposure settings.58 As with other categories discussed in this article, the key issue is not necessarily a single exposure, but the potential for repeated or cumulative exposure across multiple products and pathways.
Given the wide variability within this class of compounds, evaluating individual ingredients—rather than assuming all glycol ethers behave similarly—is an important part of understanding potential risk. However, my focus here is on other glycol ethers rather than propylene glycol.
Human epidemiological research has raised concerns about certain glycol ethers and male reproductive health. One large study involving more than 2,000 participants reported that men with occupational exposure to glycol ethers had substantially lower sperm counts than men without such exposure, even after controlling for other variables.59
Another epidemiological study examining 2-ethoxyethanol (2-EE) and 2-methoxyethanol (2-ME) likewise found increased rates of oligospermia and azoospermia, both of which reflect impaired sperm production.60 While these specific glycol ethers are not commonly used in personal care products, they are used in other consumer and industrial applications, and their documented effects are important when evaluating the broader safety of this chemical class.
This raises a reasonable and important question: whether other glycol ethers with similar chemical structures—such as phenoxyethanol, which is commonly used in personal care products—may produce similar biological effects. At present, there is limited research directly addressing this question. That lack of data should not be interpreted as evidence of safety, but rather as a gap in the research that has yet to be fully explored.
Additional research has further examined the reproductive effects of glycol ethers. Review papers summarizing toxicological data have outlined multiple lines of evidence linking certain glycol ethers to reproductive toxicity and have proposed mechanisms involving disruption of cellular and hormonal processes.58
Case-control studies in human populations have also reported associations between exposure to specific glycol ethers and adverse reproductive outcomes in men, adding to the evidence observed in occupational settings.60
Animal studies provide additional insight into potential developmental effects. For example, research in rodents has shown that exposure of pregnant females to certain glycol ethers can result in measurable changes in male offspring, including reduced testicular weight.61 While experimental exposure conditions may differ from everyday use, these findings are relevant when considering real-world scenarios involving repeated or prolonged exposure—such as occupational settings or frequent use of products containing these compounds.
 Oxybenzone, a common sunscreen active ingredient.
Oxybenzone, a common sunscreen active ingredient.
5+. Sunscreen Ingredients: Avobenzone, Octocrylene, Homosalate, and Oxybenzone
These ingredients are commonly used as “UV filters” in a wide range of products marketed for sun protection. Unlike mineral sunscreens (such as zinc oxide), these compounds are designed to absorb UV radiation through chemical reactions in the skin.

As discussed in more detail in a separate article, a number of these compounds have been studied for their potential to interact with hormone systems. Research has shown that some of these ingredients exhibit endocrine-disrupting activity, including effects on androgen and estrogen signaling pathways.24,50,51
For example, avobenzone—one of the most widely used non-mineral UV filters—has demonstrated anti-androgen activity in laboratory (in vitro) studies, indicating that it may interfere with normal male hormone signaling.50 Similarly, animal studies have shown that octocrylene can exhibit both anti-androgenic and anti-estrogenic activity, suggesting disruption of normal hormone balance in multiple directions.51
Other research has found that homosalate and related UV filter compounds can act as antagonists of the androgen receptor, meaning they block the normal action of androgens such as testosterone. In addition, some of these compounds have demonstrated interactions with estrogen receptors as well, further reinforcing that their biological activity is not limited to a single hormonal pathway.24
Taken together, these findings indicate that certain commonly used sunscreen ingredients are biologically active and capable of interfering with hormone signaling. While the extent of real-world impact continues to be studied, their widespread use and repeated application make them relevant when considering cumulative exposure to endocrine-active compounds. Oxybenzone (also known as benzophenone-3) has been shown in experimental studies to exhibit estrogenic activity and to affect reproductive endpoints in animal models.24,51
Bringing this discussion to human data, population-based studies have also examined associations between oxybenzone exposure and hormone levels. Analysis of CDC-collected data from the National Health and Nutrition Examination Survey (NHANES) found that higher levels of benzophenone-3 (BP-3) in urine were associated with lower total testosterone levels in male adolescents.62 The same study also reported associations between other endocrine-disrupting compounds and altered hormone levels in both males and females.
These findings are important because they demonstrate that exposure to certain commonly used chemical ingredients is measurable in human populations and is associated with detectable changes in hormone levels. While association does not prove causation, the consistency between experimental and human data strengthens the concern that these compounds may influence endocrine function.
 Tartrazine, AKA “FD&C yellow 5”.
Tartrazine, AKA “FD&C yellow 5”.
6. Tartrazine (FD&C Yellow 5)
artrazine is a synthetic colorant widely used in both personal care products and processed foods. It is also commonly used as a pharmaceutical color additive. It should not be confused with atrazine, a separate chemical used as a pesticide, which has been studied for its own endocrine-disrupting effects. Because of its widespread use, exposure to tartrazine can occur through multiple pathways, including ingestion and topical application.

Although the volume of research on tartrazine is more limited compared to some other compounds discussed in this article, the available data raises legitimate concerns. Experimental studies in animal models have reported adverse effects following exposure, including reduced testicular weight, decreased sperm counts, abnormalities in sperm morphology, and changes in body weight.63
These findings are particularly relevant given how frequently tartrazine appears in everyday products. While additional research is needed to fully characterize its effects in humans, the existing data indicates that it is not biologically inert. As with many compounds discussed here, the combination of widespread exposure and evidence of biological activity warrants careful consideration. A common argument is that long-term use of a chemical implies safety—that if meaningful harm existed, it would already be widely recognized. However, this assumption does not account for the complexity of detecting subtle or long-term biological effects, particularly when exposures are widespread and occur over extended periods. Absence of widespread recognition is not the same as absence of risk.
Additional experimental studies on tartrazine provide further insight. While not all studies report identical findings, some have confirmed adverse effects on male reproductive parameters, including reduced sperm counts and abnormalities in sperm motility and structure, even when other measures such as body or testes weight were unchanged.63
Mechanistic studies have also explored how tartrazine may exert these effects. In vitro research has demonstrated that tartrazine can exhibit estrogen-like (xenoestrogenic) activity, even at relatively low concentrations, providing a potential explanation for the reproductive findings observed in animal models.63 Additional mechanistic research provides further context for the findings observed in animal studies. Some studies have reported that tartrazine exposure is associated with reduced levels of antioxidant enzymes in testicular tissue, which may increase susceptibility to oxidative stress. This effect has been linked to the potential generation of reactive oxygen species (ROS), which can contribute to cellular damage when not adequately controlled.63
Beyond reproductive effects, animal studies have also reported impacts on other organ systems. For example, changes in markers of liver and kidney function—including ALT, AST, ALP, urea, creatinine, total protein, and albumin—have been observed following tartrazine exposure.63 Additional studies have confirmed alterations in liver structure (histopathology), further supporting evidence of systemic effects beyond the reproductive system.
In vitro research has also explored the potential for genotoxic effects, suggesting that tartrazine may interact with genetic material under certain conditions. However, these findings have not yet been consistently demonstrated in animal models, and further research is needed to clarify their relevance.
Taken together, the available evidence indicates that tartrazine is biologically active and capable of affecting multiple systems. While the current body of research is more limited compared to some other compounds discussed in this article, the findings that do exist raise valid concerns—particularly given the frequency of exposure. As a result, individuals seeking to minimize unnecessary exposure may reasonably choose to avoid its use where practical.
Section 4
Where These Ingredients Are Commonly Found
At this point, the most practical question becomes: where are these ingredients actually found?
Rather than focusing on any single manufacturer, it is more important to understand that the ingredients discussed in this article are widely used across the personal care and household product industries. They can be identified directly by reading ingredient labels.
Common product categories where these compounds are frequently encountered include:
• Body washes, shampoos, conditioners, and deodorants (often containing “fragrance/parfum”)
• Lotions, creams, and cosmetics (may contain parabens or glycol ethers)
• Sunscreens and moisturizers with UV filters (such as oxybenzone, octocrylene, or avobenzone)
• Laundry detergents and fabric softeners (often containing fragrance-related compounds)
• Dishwashing liquids and household cleaners (which may contain glycol ethers and colorants)
• Processed foods and pharmaceuticals (which may contain colorants such as tartrazine)
Because many of these ingredients are not always individually disclosed—particularly when listed under general terms like “fragrance”—careful label review is essential. In some cases, identifying exposure requires cross-referencing ingredients with independent safety databases or scientific literature.
The key takeaway is that exposure does not come from a single product, but from repeated use of multiple products over time. This cumulative exposure is what makes ingredient awareness and informed decision-making especially important.

Examples from commonly available product ingredient lists further illustrate how these compounds appear in everyday use.
Fragranced personal care products such as body washes, shampoos, and deodorants frequently list “fragrance” or “parfum,” which may include phthalates. Some of these products also contain additional ingredients such as sodium laureth sulfate and sodium lauryl sulfate, which can increase skin permeability and potentially enhance absorption of other compounds.
Certain shampoos and personal care products also contain preservatives such as methylchloroisothiazolinone and methylisothiazolinone, which are known skin irritants. In some cases, formulations may include both sodium benzoate and acids such as citric acid or ascorbic acid—combinations that have been studied for their potential to form benzene under specific conditions.
Laundry detergents, fabric softeners, and household cleaning products commonly contain fragrance-related compounds as well as solvents such as glycol ethers. Because these products are used frequently and come into contact with clothing, dishes, and surfaces, they can contribute to ongoing exposure.
Even products marketed as “natural” or “gentle” may still contain synthetic preservatives, fragrances, or other compounds discussed in this article. For this reason, marketing claims should not be relied upon as a substitute for reviewing ingredient lists.

Household products such as dishwashing liquids, shampoos, and hair care products provide additional exposure pathways to the compounds discussed in this article.
Dishwashing liquids, for example, may contain fragrance-related compounds, glycol ethers, and colorants. Because these products are used directly on surfaces that come into contact with food, even small amounts of residue that remain after rinsing may contribute to ingestion over time.
Similarly, hair care products—including shampoos and conditioners—often contain combinations of fragrance, preservatives, surfactants, and other additives. Some of these ingredients, such as methylchloroisothiazolinone and methylisothiazolinone, are known skin irritants, while others may increase skin permeability, potentially enhancing absorption of additional compounds.
It is also common for these products to contain multiple classes of ingredients discussed earlier in this article, including fragrance components, glycol ethers, and colorants. Because these products are used regularly and over large surface areas of the body, they can represent a consistent source of exposure.
The key issue is not any single product, but the repeated and combined use of many such products over time, which may contribute to cumulative exposure through both dermal contact and incidental ingestion.

An important challenge in evaluating product safety is the lack of full ingredient transparency in certain formulations—particularly those that include “fragrance” or similar proprietary blends. While some products or brands may state that they do not use specific compounds such as phthalates, this is not always consistently disclosed across all product lines or categories.
This inconsistency highlights a broader issue: without full disclosure of all components within proprietary formulations, it can be difficult for consumers to independently verify what is—and is not—present in a given product. As a result, uncertainty remains regarding potential exposure to certain compounds, especially those known to be commonly associated with fragrance mixtures.
In situations where complete ingredient transparency is not available, consumers are left to make decisions based on partial information. For this reason, some individuals choose to limit or avoid products that rely on undisclosed fragrance components, or prioritize products that provide full ingredient disclosure.

As discussed throughout this article, the examples provided represent only a small sampling of the types of ingredients commonly found in personal care and household products. Because these ingredients are used across many product categories, exposure is not limited to a single item—it is cumulative, occurring through repeated daily use over time.
At a certain point, attempting to manage exposure on a product-by-product basis becomes impractical. When ingredient transparency is limited and compounds of concern continue to appear across a wide range of commonly used products, some consumers choose a more decisive response: not just avoiding these products, but actively boycotting them.
A boycott is a way for consumers to exercise their voice in the marketplace. Choosing not to purchase products that rely on undisclosed fragrance systems or ingredients with concerning biological activity sends a clear signal about what is—and is not—acceptable. It is a direct and practical response to the combination of widespread exposure, incomplete disclosure, and the body of scientific evidence discussed throughout this article.
If you choose to continue using such products, careful review of ingredient labels and independent research remains essential. But for those who prefer a clearer and more consistent approach, boycotting these ingredient categories—and supporting companies that prioritize full transparency—offers a straightforward path forward.

At the end of the day, this comes down to awareness and choice. The research is available, the ingredients are listed—at least in part—and the patterns are clear enough for individuals to make informed decisions. You do not need perfect information to take action. If the level of uncertainty, cumulative exposure, and lack of transparency outlined in this article concerns you, then choosing to boycott products that rely on these ingredients is a reasonable and proactive step. Where you spend your money matters. Every purchase either reinforces the status quo or supports change. Choose accordingly.
Rob
References:
1. Gore AC, Chappell VA, Fenton SE, et al. EDC-2: The Endocrine Society’s Second Scientific Statement on Endocrine-Disrupting Chemicals. Endocr Rev. 2015;36(6):E1–E150.
2. Diamanti-Kandarakis E, Bourguignon JP, Giudice LC, et al. Endocrine-disrupting chemicals: an Endocrine Society scientific statement. Endocr Rev. 2009;30(4):293–342.
3. Skakkebaek NE, Rajpert-De Meyts E, Buck Louis GM, et al. Male reproductive disorders and fertility trends: influences of environment and genetic susceptibility. Physiol Rev. 2016;96(1):55–97.
4. Meeker JD, Sathyanarayana S, Swan SH. Phthalates and other additives in plastics: human exposure and associated health outcomes. Philos Trans R Soc Lond B Biol Sci. 2009;364(1526):2097–2113.
5. Boberg J, Taxvig C, Christiansen S, Hass U. Possible endocrine disrupting effects of parabens and their metabolites. Reprod Toxicol. 2010;30(2):301–312.
6. Rochester JR, Bolden AL. Bisphenol S and F: A systematic review and comparison of hormonal activity. Environ Health Perspect. 2015;123(7):643–650.
7. Swan SH. Environmental phthalate exposure in relation to reproductive outcomes and other health endpoints in humans. Environ Res. 2008;108(2):177–184.
8. Bergman Å, Heindel JJ, Jobling S, Kidd KA, Zoeller RT. State of the science of endocrine disrupting chemicals. World Health Organization / United Nations Environment Programme. 2013.
9. Sharpe RM, Skakkebaek NE. Testicular dysgenesis syndrome: mechanistic insights and potential new downstream effects. Fertil Steril. 2008;89(2 Suppl):e33–e38.
10. Sharpe RM. Environmental/lifestyle effects on spermatogenesis. Philos Trans R Soc Lond B Biol Sci. 2010;365(1546):1697–1712.
11. Skakkebaek NE, Rajpert-De Meyts E, Main KM. Testicular dysgenesis syndrome: an increasingly common developmental disorder with environmental aspects. Hum Reprod. 2001;16(5):972–978.
12. Toppari J, Virtanen HE, Main KM, Skakkebaek NE. Cryptorchidism and hypospadias as a sm number and motile activity on the F1 offspring maternally exposed to butyl p-hydroxybenzoic acid (butylparaben). J Vet Med Sci. 2002;64(3):227–235.ign of testicular dysgenesis syndrome (TDS): environmental connection. Birth Defects Res A Clin Mol Teratol. 2010;88roid hormone levels: results (10):910–919.
13. Zoeller RT, Brown TR, Doan LL, et al. Endocrine-disrupting chemicals and public health protection: a statement of principles from The Endocrine Society. Endocrinology. 2012;153(9):4097–4110.
14. Levine H, Jørgensen N, Martino-Andrade A, et al. Temporal trends in sperm count: a systematic review and meta-regression analysis. Hum Reprod Update. 2017;23(6):646–659.
15. Travison TG, Araujo AB, O’Donnell AB, Kupelian V, McKinlay JB. A population-level decline in serum testosterone levels in American men. J Clin Endocrinol Metab. 2007;92(1):196–202.
18. Massart F, Parrino R, Seppia P, Federico G, Saggese G. How do environmental estrogen disruptors induce precocious puberty? Minerva Pediatr. 2006;58(3):247–254.
19. Bodicoat DH, Schoemaker MJ, Jones ME, et al. Timing of pubertal stages and breast cancer risk: the Breakthrough Generations Study. Breast Cancer Res. 2014;16(1):R18.
20. Durmaz E, Özmert EN, Erkekoglu P, et al. Plasma bisphenol A levels in girls with central precocious puberty. J Clin Res Pediatr Endocrinol. 2014;6(1):16–21.21. Yager JD, Davidson NE. Estrogen carcinogenesis in breast cancer. N Engl J Med. 2006;354(3):270–282.
22. Soto AM, Sonnenschein C. Environmental causes of cancer: endocrine disruptors as carcinogens. Nat Rev Endocrinol. 2010;6(7):363–370.
23. Darbre PD, Harvey PW. Paraben esters: review of recent studies of endocrine toxicity, absorption, esterase and human exposure, and discussion of potential human health risks. J Appl Toxicol. 2008;28(5):561–578.
24. Wetherill YB, Akingbemi BT, Kanno J, et al. In vitro molecular mechanisms of bisphenol A action. Reprod Toxicol. 2007;24(2):178–198.
26. Dodson RE, Nishioka M, Standley LJ, et al. Endocrine disruptors and asthma-associated chemicals in consumer products. Environ Health Perspect. 2012;120(7):935–943.
28. James-Todd T, Terry MB, Rich-Edwards J, Deierlein A, Senie R. Childhood hair product use and earlier age at menarche in a racially diverse study population. Ann Epidemiol. 2011;21(6):461–465.
29. Eberle CE, Sandler DP, Taylor KW, White AJ. Hair dye and chemical straightener use and breast cancer risk in a large US population of black and white women. Int J Cancer. 2020;147(2):383–391.
31. Ho SM, Tang WY, Belmonte de Frausto J, Prins GS. Developmental exposure to estradiol and bisphenol A increases susceptibility to prostate carcinogenesis and epigenetic alterations. Carcinogenesis. 2006;27(3):583–590.
32. Berry SJ, Coffey DS, Walsh PC, Ewing LL. The development of human benign prostatic hyperplasia with age. J Urol. 1984;132(3):474–479.
34. Bosland MC. The role of estrogens in prostate carcinogenesis: a rationale for chemoprevention. Rev Urol. 2009;11(2):84–93.
38. Health Canada. Phthalates in cosmetic and personal care products: concentrations and possible dermal exposure. Government of Canada; 2016.
40. Swan SH, Main KM, Liu F, et al. Decrease in anogenital distance among male infants with prenatal phthalate exposure. Environ Health Perspect. 2005;113(8):1056–1061.
63. Amin KA, Hameid HA, Abd Elsttar AH. Effect of food azo dye tartrazine on some biochemical parameters related to renal, hepatic function and oxidative stress in mice. Food Chem Toxicol. 2010;48(10):2994–2999.
Nature's Complement is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program. If you purchase products on Amazon through any of our affiliate links, we get a small percentage of the transaction, at no extra cost to you. We spend a lot of time writing the articles on this site, and all this information is provided free of charge. When you use our affiliate links, you support the writing you enjoy without necessarily buying our products. (However we would appreciate if you would do that too!) Thank you for helping to support our work, however you choose to do so.
These statements have not been evaluated by the Food and Drug Administration. This information and/or products are not intended to diagnose, treat, cure or prevent any disease.


